CRP is a dedicated clinical research site at the forefront of clinical research, partnering with industry leaders to improve patient outcomes.
Our staff and our enrollment reflect the community we serve. See the breakdown of who's enrolled in our trials this past year below.
Our fully electronic Clinical Trial system allows us to minimize administrative burden and streamline operations so our team can focus more on patient engagement and protocol adherence.
Our participants are the reason we come to work every day. Our decisions and daily processes are guided by the principle that participants will always be the heart of our work.
We are proud of our committed research team, most of whom have been with our site for over 15 years. Our cohesiveness as a team and our collective experiences enable us to develop innovative solutions for the complex needs of our clinical trials.
Aggregate participant demographics across all studies, trailing 12 months. Pulled live from our clinical trial management system.
Demographics pulled live from our clinical trial management system. Updated daily.
Every participant gets free rideshare to and from study visits. No car needed, no cost to you.
Participants receive payment for every visit, plus access to investigational treatments at no cost.
State-of-the-art facilities in Philadelphia and Pennington, NJ with free parking and flexible scheduling.
State-of-the-art research sites in Philadelphia, PA and Pennington, NJ — both staffed and equipped to handle Phase II–IV trials end-to-end, with dedicated sponsor and monitor workspace.
Fully electronic CTMS (CRIO) plus a custom BigQuery and agent-driven analytics layer powering live dashboards. No paper trail to chase, no double entry.
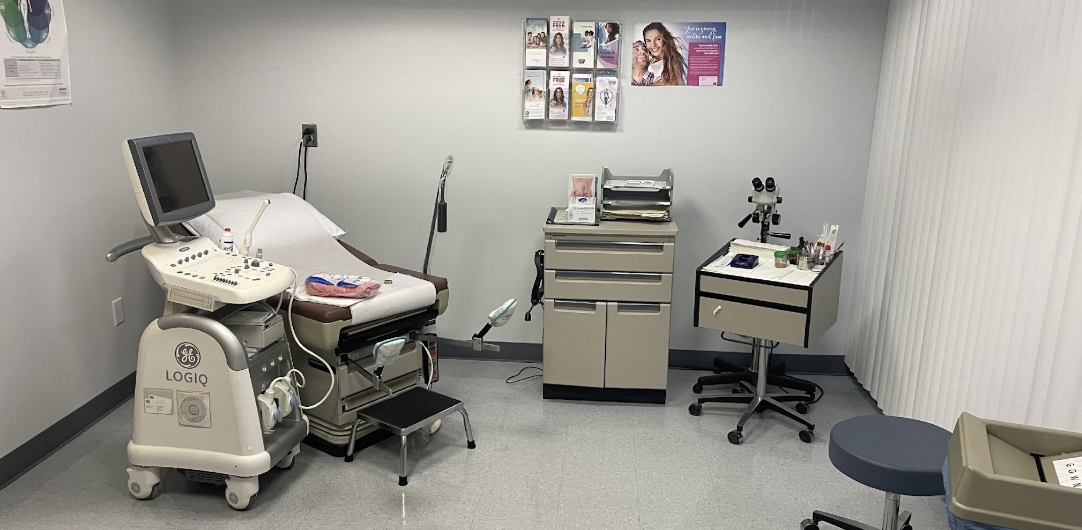
FibroScan, ultrasound, centrifuges, EKGs, infusion pumps, and incubators on premise — most studies need no off-site vendors.
Temperature-controlled ambient, refrigerated (2–8°C), and frozen (-20°C / -80°C) investigational product storage with 24/7 alerting.
Private exam rooms, on-campus urgent care, free parking at both sites, and free rideshare to and from every visit for all participants.
Our team mirrors the patient populations we serve, building trust with participants and reducing screen failure and dropout.
Dedicated monitor workstations, sponsor conference and training room, private investigator and coordinator offices, and a sample processing & shipping lab.


























All studies conducted under FDA oversight with Investigational New Drug authorization.
Every protocol reviewed and approved by an independent Institutional Review Board before enrollment begins.
Patient data protected under strict HIPAA privacy and security standards at every stage of the research process.
Get to know the dedicated team at Clinical Research Philadelphia.